Christian Doppler Laboratory for Next Generation CAR T Cells
T cells engineered to express Chimeric Antigen Receptors (CAR T cells) have shown impressive clinical success for patients with B cell malignancies. However, since CAR T cells are self-replicating living drugs it is difficult to regulate their function after administration to a patient, often resulting in severe side effects such as cytokine release syndrome and neurotoxicity. At the same time, currently used CAR T cells could also potentially attack healthy tissue, since their typical target antigens are always present to some extent on a small fraction of healthy cells. This lack of tumor specificity and the insufficient controllability of CAR T cell function are major hurdles for the clinical implementation of the full potential of CAR T cell therapy until today.
The goal of this “CD Laboratory for Next Generation CAR T cells” is therefore to generate novel molecular tools to minimize the destruction of healthy tissue and to be able to reversibly control CAR T cell activity in the patient.
- The lack of reversible control of CAR T cell activation is addressed by constructing molecular ON-switches that can be controlled by administration of approved, orally available drugs. These novel molecular switches can not only be used to regulate CAR T cell function but will open up a completely new realm of cell engineering.
- The lack of tumor specificity of CAR T cells is addressed by constructing a novel class of AND gate CARs for combinatorial antigen recognition. That is, those CARs are only activated if both antigens A AND B are present on a target cell, but not if only one antigen is expressed. If those two antigens are selected accordingly, this specificity for double-positive cells can greatly improve tumor specificity.
Our CD Laboratory will provide a comprehensive set of novel approaches, in which an essential principle is the use of proteins and small molecules that allow for rapid clinical implementation. Our multidisciplinary approach will enable the generation and clinical translation of novel CAR T cell therapies with improved efficacy, safety and tumor specificity.

F. l. t. r.: Thomas Gabler, Mara Mitstorfer, Charlotte Brey, Manfred Lehner, Magdalena Teufl, Elise Sylvander, Michael Traxlmayr, Benjamin Salzer
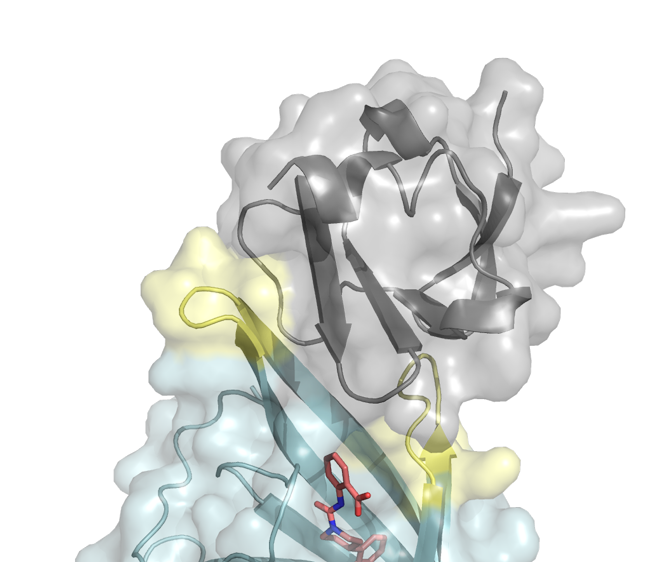
New molecular tools for improving safety and specificity of CAR T cell therapy.
News
(November 2023): For Women in Science – Prestigious scholarship for Charlotte Zajc
(Vienna, 27.11.2023) Charlotte Zajc, Postdoctoral Researcher from the University of Natural Resources and Life Sciences Vienna is one of the four scientists who were awarded the "For Women in Science" scholarship worth 25,000 euros on November 22!
The funding program is a cooperation between L'Oreal Austria, the Austrian UNESCO Commission and the Austrian Academy of Sciences and is financially supported by the Federal Ministry of Education, Science and Research. The scholarships enable promising female talents, who are also role models for girls and women with scientific ambitions, to advance their scientific careers. The aim is also to raise public awareness of the importance of basic research and the urgency of harnessing the potential of female scientists.

(May 2022): Elise Sylvander earns prestigious DOC fellowship
Congratulations to Elise Sylvander on receiving a DOC fellowship of the Austrian Academy of Sciences. The aim of her fellowship is to (further) develop a molecular on-switch allowing to activate therapeutic CAR T cells, once they have been administered to the patient.
“CAR T cells are very promising cancer therapies. Unfortunately, they can have strong side effects. The novelty of our CAR T cell system is that it can be switched on, employing a small molecule that demonstrates amazing pharmacodynamics and is very well tolerated, even in children. Only as long as this small molecule is taken, the CAR T cells are active”, explains Elise Sylvander.
The scientist pursues her research in the Christian Doppler Laboratory (CD-lab) for Next Generation CAR T cells under the supervision of Manfred Lehner (head, CD-lab) at St. Anna Children's Cancer Research Institute (CCRI) and Michael Traxlmayr (head of the external module) at the University of Natural Resources and Life Sciences, Vienna (BOKU).
During her two-year fellowship, Elise Sylvander is analyzing the new system in T-cells in the lab, with promising preliminary results. “Later it can be tested in mice. However, it always takes a lot of time for those kind of therapies until they can be applied in humans.”
The DOC Fellowship Program of the Austrian Academy of Sciences offers funding for highly qualified doctoral candidates in all areas of research. The fellowships allow young researchers to dedicate themselves to working in focused fashion to complete their doctoral thesis within a clear timeframe.

The grant:
Regulating CAR T cells with a safe and naturally occurring drug
CCRI responsible researcher: Elise Sylvander (supervisor: Manfred Lehner)
Grant from the Austria Academy of Sciences (ÖAW), DOC fellowship, ID – 26323
Duration: 01/07/2022 to 01/07/2024
(Mar. 2022): Interview with Charlotte Zajc in "Die Presse, Magazin"
Charlotte Zajc from the BOKU-module of St. Anna Children's Cancer Research Institute´s CD-Lab explains her research in "Die Presse". She hopes the molecular on/off-switch she helped develop will eventually be used to benefit patients: "The current study is the cornerstone. The principle works, it's feasible and efficient enough. But there is still a long way to go."
Until her recent maternity leave, Charlotte was working in the CD lab’s external module, headed by DI Dr. Michael Traxlmayr, at the University of Natural Resources and Life Sciences, Vienna (BOKU).
Full article >> Der richtige Schalter gegen Krebs | DiePresse.com
Foto: Dr. Charlotte Zajc, (c) Die Presse/Clemens Fabry

(Mar. 2022): Benjamin Salzer receives BOKU Talent Award
????CONGRATS to Benjamin Salzer, PhD, who received the BOKU Talent Award from the University of Natural Resources and Life Sciences together with the City of Vienna Anniversary Fund.
????This award recognized his outstanding scientific thesis in the thematic framework of BOKU competence fields.
????Until winter 2021, Benjamin Salzer was a Postdoctoral Fellow in the CD Laboratory for Next Generation CAR T Cells (Head: Manfred Lehner, PhD) of St. Anna Children's Cancer Research Institute (CCRI).
Photo: Dr. Benjamin Salzer (5th from left), further awardees and prize committee, (c) University of Natural Resources and Life Sciences

(Feb. 2022): Interview with Benjamin Salzer published in "Der Standard", series "Geistesblitz"
St. Anna Children's Cancer Research Institute´s CD-Lab alumni Benjamin Salzer in the Standard interview: "I was fascinated by the approach of using the immune system of patients in the fight against cancer!"
Full article >> https://www.derstandard.at/story/2000133217235/st-anna-kinderkrebsforschung-steuerung-fuer-die-krebskiller

Ben Salzer at work, copyright: Gilbert Novy
(Oct. 2021): Charlotte Zajc receives both BOKU Talent Award and BOKU Best Paper Award!
????????????⚕️CONGRATS to Charlotte Zajc, PhD, who is honored twice! The scientist from our Christian Doppler (CD) Laboratory for Next Generation CAR T cells receives the BOKU Talent Award and the BOKU Best Paper Award from the City of Vienna Jubilee Funds.
????During her PhD studies, Charlotte Zajc designed a molecular ON-switch for CAR T cells in order to control their anti-cancer activity and to reduce the risk of CAR T cells attacking healthy tissue. Her PhD thesis and the resulting publication are honored with the BOKU Best Paper Award and the BOKU Talent Award respectively.
????Charlotte Zajc is a Postdoctoral Fellow in the CD Laboratory for Next Generation CAR T Cells (Head: Manfred Lehner, PhD) of St. Anna Children's Cancer Research Institute (CCRI). She is working in the CD lab’s external module, headed by DI Dr. Michael Traxlmayr, at the University of Natural Resources and Life Sciences, Vienna (BOKU).

(Sept. 2021): Benjamin Salzer and Charlotte Zajc win first place at the Life Science Research Awards Austria 2021
????CONGRATS to Benjamin Salzer, PhD, and Charlotte Zajc, PhD, from our Christian Doppler Laboratory for Next Generation CAR T Cells! Both have won with their publication resulting from their doctoral thesis a first place at the Life Science Research Awards Austria 2021 – Benjamin Salzer (St. Anna Children's Cancer Research Institute (CCRI)) in the category Applied Research, and Charlotte Zajc (University of Natural Resources and Life Sciences, Vienna (BOKU)/St. Anna CCRI alumna) in the category Excellence and Societal Impact.
In the award-winning publications, Charlotte Zajc, Benjamin Salzer, and colleagues designed novel molecular switches and Chimeric Antigen Receptor (CAR) prototypes. With these new tools, immune cells can be reliably switched on and off and directed more specifically against tumor cells. This reduces the risk of CAR T cells attacking healthy tissue.
The official Award Ceremony took place during the 13th Annual Meeting of the ÖGMBT - Austrian Association of Molecular Life Sciences and Biotechnology on Monday, September 20. The award is supported by the Bundesministerium für Digitalisierung und Wirtschaftsstandort.
Benjamin Salzer and Charlotte Zajc both conducted their doctoral thesis at St. Anna CCRI and are Postdoctoral Fellows in the Christian Doppler Laboratory for Next Generation CAR T Cells (Head: Manfred Lehner, PhD) at St. Anna CCRI and at its External Module at BOKU Vienna (Head: DI Dr. Michael Traxlmayr), respectively.

F.l.t.r.: Benjamin Salzer, Charlotte Zajc, Photo: ÖGMBT/Philipp Schuster
(Dec. 2020): Partners from CD-Lab module at BOKU win Invention of the Year 2020 Award
Each year, the University of Natural Resources and Life Sciences (BOKU), Vienna awards the most outstanding invention. In 2020, the prize went to our co-inventors from the external module of CCRI´s Christian-Doppler Laboratory for Next Generation CAR T Cells, Michael Traxlmayr and Charlotte Zajc with the technology "A lipocalin fold-based inducible dimerization system ("Lipocalin-Switch")".
This invention is the result of an excellent scientific cooperation with our research partners from the BOKU, as well as our industry partners from the company Miltenyi Biotec. It is characterized in particular by the wide range of possible applications, which makes its exploitation possible in many areas.
The CD Laboratory aims to enable safe clinical translation of more effective therapies and to provide a major boost to cancer therapy worldwide. The awarded invention is the basis for the exploration of new strategies to improve the safety of CAR T cells, and thus could play a key role in unfolding the full potential of CAR T cell therapy.
Link to the award ceremony at BOKU: https://boku.ac.at/oeffentlichkeitsarbeit/news-container/unsere-wissenschaftlichen-visionaerinnen-und-wegbereiterinnen
Learn more about BOKU: https://boku.ac.at/
Photo (f.l.t.r.): Daniel Paul Komuczki, Gordana Wozniak-Knopp, David Brunmayr (Organic Tools), Isabella Zangl, Christian Obinger, Michael Traxlmayr, Charlotte Zajc, Michael Moll (accent), Maria Georgiades, Raphaela Hellmayr
© BOKU Medienstelle/Christoph Gruber

(Sept. 2020): CD-Lab Head Manfred Lehner comments on CAR T cell research in the journal Cell Systems
We shouldn´t fear complexity
Manfred Lehner, head of CCRI´s Christian Doppler Laboratory for Next Generation Car T Cells was invited to offer his short opinion on CAR T cell research in the article format “Voices” from the renowned scientific journal Cell Systems (Volume 11, September 23, 2020, https://pubmed.ncbi.nlm.nih.gov/32970992/). Together with other experts in the field, he comments on what the optimal design-build-test cycle for clinically relevant synthetic CAR T cell therapies could be.
By comparing with the breathtaking IT development from the early 1960s until now, where technological innovation has made computers much smaller but far more complex and powerful, Lehner explains that the challenge in the CAR T cell development is of similar ambivalence: “We have to strongly increase the efficacy of CAR T cells and at the same time reduce their potential toxicity. To fully unfold the potential of CAR T cells, complexity will certainly increase. However, we should not be afraid of it in our attempts to find solutions, because history inspires us that something which seems impossible today might be the standard in the future.”

Picture: Excerpt from full article format "Voices", Cell Systems 11, September 23, 2020, https://pubmed.ncbi.nlm.nih.gov/32970992/
(Sept. 2020): CD-Lab Member Benjamin Salzer wins Life Science PhD Award from ÖGMBT
 CONGRATULATIONS to Benjamin Salzer. He wins the Life Science PHD Awards Austria 2020 in the category Applied Science for his outstanding thesis "Safety and specificity engineering of chimeric antigen receptor T cells (CAR-T cells) by exploitation of avidity".
CONGRATULATIONS to Benjamin Salzer. He wins the Life Science PHD Awards Austria 2020 in the category Applied Science for his outstanding thesis "Safety and specificity engineering of chimeric antigen receptor T cells (CAR-T cells) by exploitation of avidity".
After the official Award Ceremony during the 12th ÖGMBT - Austrian Association of Molecular Life Sciences and Biotechnology Annual Meeting Salzer presents his award-winning work. "Our avidity-controlled CARs depend on bivalent antigen binding and are based on two principles: controlled dimerization of two CAR subunits and antigen binding with low affinity", explains Salzer.
The prize is supported by Polymun.
St. Anna Kinderkrebsforschung (Children's Cancer Research Institute - CCRI)
https://science.ccri.at/
Photo: ÖGMBT
- Article in the Austrian newspaper "Kurier": Kurier Benjamin Salzer ÖGMBT Award
- Interview at the medical news platform MEDimpressions: https://medimpressions.at/forscherinnen-im-portraet-di-benjamin-salzer-phd/
(Aug. 2020) New review: Driving CARs with alternative navigation tools - the potential of engineered binding scaffolds
Driving CARs with alternative navigation tools - the potential of engineered binding scaffolds.
Zajc CU, Salzer B, Taft JM, Reddy ST, Lehner M, Traxlmayr MW.
FEBS J. 2020 Aug 13. doi: 10.1111/febs.15523. Online ahead of print.PMID: 32794303 Review.
(Aug. 2020) New publication: Engineering AvidCARs for combinatorial antigen recognition and reversible control of CAR function
Engineering AvidCARs for combinatorial antigen recognition and reversible control of CAR function.
B Salzer, C M Schueller, C U Zajc, T Peters, M A Schoeber, B Kovacic, M C. Buri, E Lobner, O Dushek, J Huppa, C Obinger, E M Putz, W Holter, M W Traxlmayr*, M Lehner*, Nature Communications 20th August 2020; https://rdcu.be/b6nrm, https://www.nature.com/articles/s41467-020-17970-3
(June 2020) New publication: A conformation-specific ON-switch for controlling CAR T cells with an orally available drug
A conformation-specific ON-switch for controlling CAR T cells with an orally available drug.
Zajc (neé Brey) CU., M. Dobersberger, I. Schaffner, G. Mlynek, D. Pühringer, B. Salzer, K. Djinović-Carugo, Steinberger P, De Sousa Linhares A, Yang NJ, C. Obinger, W. Holter, MW. Traxlmayr & M. Lehner.
PNAS. 2020; doi.org/10.1073/pnas.1911154117
